Every year, thousands of American laboratories handle millions of diagnostic tests that directly impact patient care. In an industry where even a small mistake can have major consequences, setting up a reliable workflow matters more than ever. This step-by-step guide walks you through each critical stage from preparing the environment to delivering results, helping American labs maintain accuracy, compliance, and trust at every point. Laboratory errors affect up to 13 percent of all diagnostic tests making attention to detail essential for quality outcomes.
Table of Contents
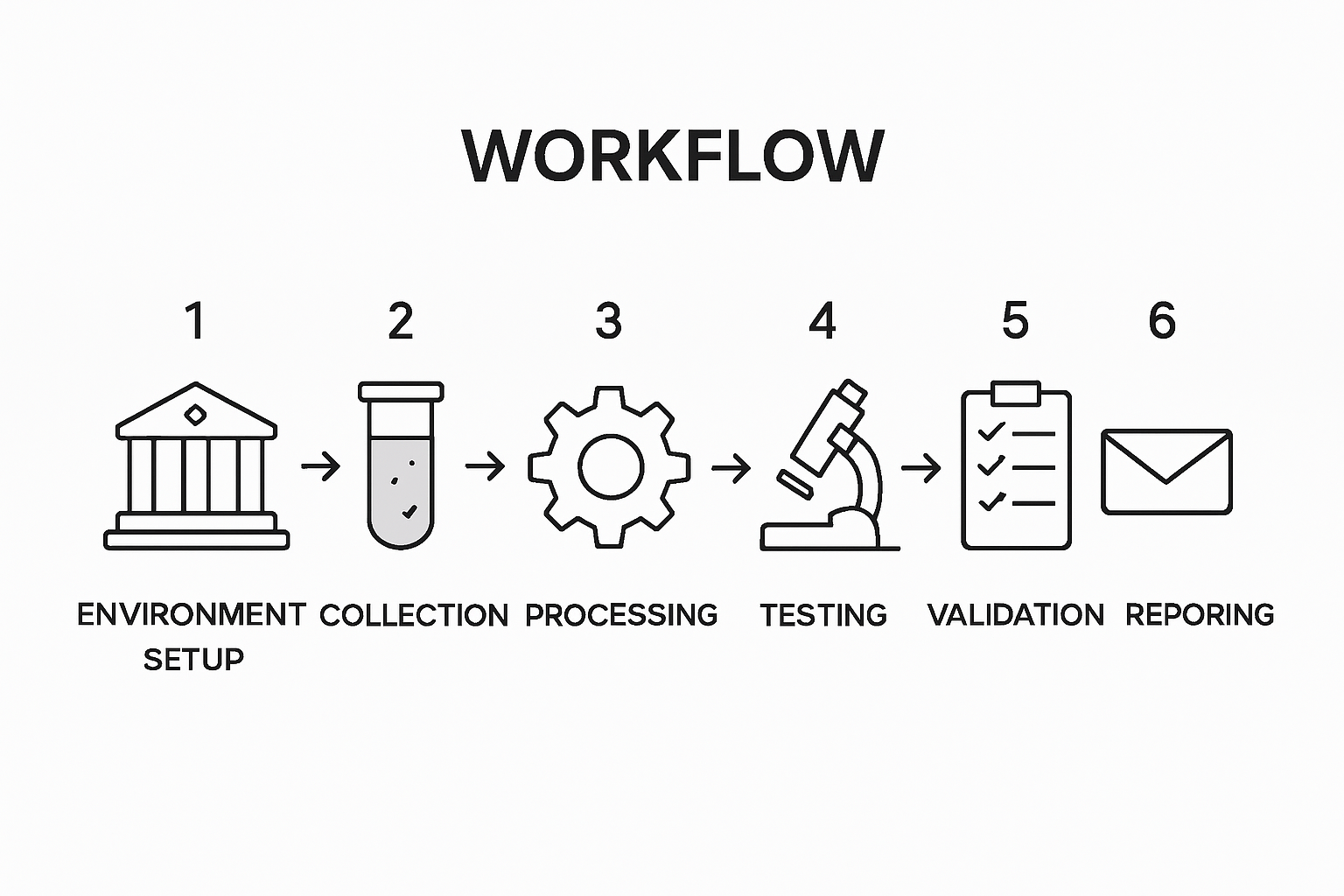
- Step 1: Set Up Diagnostic Testing Environment
- Step 2: Collect and Label Patient Samples
- Step 3: Process and Prepare Samples for Analysis
- Step 4: Conduct Diagnostic Testing Procedures
- Step 5: Validate Results and Ensure Quality
- Step 6: Report Findings to Healthcare Providers
Quick Summary
| Key Point | Explanation |
|---|---|
| 1. Establish a Clean Testing Environment | Designate an organized workspace with proper ventilation to ensure accurate testing results and maintain safety standards. |
| 2. Implement Standardized Sample Collection | Use verified labeling protocols and checklists to ensure accurate patient identification and sample integrity throughout the collection process. |
| 3. Follow Precise Sample Processing Protocols | Maintain diagnostic accuracy by adhering to standardized procedures during sample preparation and processing to prevent errors. |
| 4. Conduct Rigorous Testing Procedures | Calibrate equipment and document every step meticulously to guarantee reliable results and aid in troubleshooting. |
| 5. Create Detailed Reporting for Healthcare Providers | Construct clear and comprehensive reports that translate raw data into clinically relevant insights to assist in patient care decisions. |
Step 1: Set Up Diagnostic Testing Environment
Creating a reliable diagnostic testing environment requires meticulous planning and adherence to strict quality management principles. Your goal is to establish a space that guarantees accurate testing results while maintaining the highest standards of safety and compliance.
Starting with facility design, you will need to consider several key elements. First, designate a clean, organized workspace with proper ventilation and controlled temperature conditions. Laboratory certification requirements mandate specific environmental controls to prevent contamination and ensure precision. This means investing in appropriate storage solutions for testing materials, creating designated areas for different testing stages, and implementing strict protocols for equipment placement and handling.
Pro tip: Always develop a comprehensive standard operating procedure (SOP) document that outlines every step of your testing workflow. This not only helps maintain consistency but also serves as a critical reference for training new staff and ensuring ongoing quality control. By meticulously setting up your diagnostic testing environment with attention to detail and regulatory compliance, you create a foundation for reliable and accurate medical testing.
Your next step will involve selecting and calibrating the specialized equipment needed for your specific diagnostic testing requirements.
Step 2: Collect and Label Patient Samples
Collecting and labeling patient samples is a critical process that demands precision and careful attention to detail. Your primary objective is to ensure accurate patient identification and sample integrity throughout the entire diagnostic testing workflow.
Begin by preparing all necessary collection materials and documentation. Patient and specimen identification protocols require you to verify patient information before starting the collection process. This means confirming the patient’s full name, date of birth, and unique identifier on all collection containers and accompanying documentation. Use permanent markers or preprinted labels that are resistant to moisture and handling. Each sample container must be clearly marked with the patient’s information, collection date and time, and any specific test requirements.
Pro tip: Create a systematic approach to sample collection by developing a standardized checklist that guides you through each step. This helps minimize human error and ensures consistent documentation across all patient samples. Remember that proper labeling is not just about writing down information its about creating a reliable chain of documentation that protects patient privacy and guarantees accurate test results.
Your next step will involve carefully packaging and transporting the collected samples to the diagnostic testing laboratory while maintaining their integrity and chain of custody.
Step 3: Process and Prepare Samples for Analysis
Processing and preparing patient samples requires a systematic and precise approach to maintain diagnostic accuracy. Your goal is to transform raw collected samples into laboratory ready specimens through standardized laboratory procedures that guarantee reliable testing results.
Begin by implementing careful sample preparation protocols. This involves centrifuging blood samples to separate components, aliquoting specimens into appropriate storage containers, and ensuring proper temperature management. Safety protocols and quality assurance practices mandate specific handling techniques to prevent sample degradation. Wear appropriate personal protective equipment like gloves and lab coats, and use sterile techniques when transferring or processing samples. Pay close attention to sample volume requirements and specific preparation instructions for different types of diagnostic tests.
Pro tip: Create a detailed tracking system that documents each sample processing step. This helps maintain a comprehensive chain of custody and ensures that every specimen can be traced from collection through analysis. By meticulously following standardized processing protocols, you minimize potential errors and protect the integrity of diagnostic testing.
Your next step will involve loading prepared samples into the appropriate diagnostic testing equipment for comprehensive analysis.
Step 4: Conduct Diagnostic Testing Procedures
Conducting diagnostic testing procedures requires precision, attention to detail, and strict adherence to federal laboratory regulations. Your primary objective is to execute testing protocols that guarantee accurate and reliable results while maintaining the highest standards of scientific integrity.
Begin by carefully calibrating your diagnostic equipment and preparing testing reagents according to manufacturer specifications. Individualized Quality Control Plans provide a systematic approach to managing potential testing errors through comprehensive risk assessment. This means performing preliminary checks on all testing instruments, verifying reagent quality, and establishing baseline performance metrics before initiating any diagnostic procedure. Follow a meticulous step by step process for each specific test type, ensuring that every sample is processed under controlled and standardized conditions.
Pro tip: Document every step of your testing procedure with extreme precision. Maintain a comprehensive log that tracks equipment performance, sample handling, and test outcomes. This not only supports quality assurance but also provides a critical reference for troubleshooting and continuous improvement in your diagnostic workflow. By implementing rigorous testing protocols and maintaining detailed documentation, you create a foundation for consistently reliable diagnostic results.
Your next step will involve carefully analyzing and interpreting the test results to generate accurate diagnostic insights.
Step 5: Validate Results and Ensure Quality
Validating diagnostic test results is a critical process that demands meticulous attention and systematic verification to guarantee the highest level of accuracy. Your primary goal is to implement comprehensive quality management strategies that thoroughly examine and confirm the reliability of each diagnostic test outcome.
Begin by cross referencing your test results with established baseline parameters and control samples. Customized quality control plans enable you to develop a targeted approach for identifying potential errors or anomalies in your testing process. This involves performing multiple validation checks, comparing results against reference ranges, and conducting statistical analysis to detect any significant deviations. Examine each result for consistency, looking for patterns that might indicate systematic errors in equipment calibration or sample preparation.
Pro tip: Implement a robust review protocol that includes multiple levels of verification. Create a comprehensive checklist that requires independent review by two different laboratory professionals before finalizing any diagnostic report. This additional layer of scrutiny helps minimize human error and provides an extra safeguard to ensure the highest possible accuracy in your diagnostic testing workflow.
Your next step will involve preparing a detailed report that communicates the validated test results to healthcare providers or patients.
Step 6: Report Findings to Healthcare Providers
Reporting diagnostic test findings is a critical communication process that bridges laboratory insights with clinical decision making. Your primary objective is to deliver timely and comprehensive laboratory results that provide healthcare providers with clear and actionable diagnostic information.
Begin by constructing a detailed report that includes not just raw data, but contextual interpretation of the test results. Effective laboratory communication requires translating technical findings into clinically meaningful insights. This means providing reference ranges, highlighting any abnormal values, and including potential clinical implications. Ensure that the report is formatted clearly with sections for patient identification, test details, results, and any critical observations that might require immediate medical attention.
Pro tip: Develop a standardized reporting template that includes a quick reference section for critical or urgent findings. This allows healthcare providers to rapidly identify and respond to potentially significant medical indicators. By creating a systematic and comprehensive reporting approach, you transform raw diagnostic data into a valuable tool for patient care and medical decision making.
Your final step will involve securely transmitting the diagnostic report to the appropriate healthcare provider while maintaining patient confidentiality.
Elevate Your Diagnostic Testing Workflow with Centra Diagnostics
Mastering the diagnostic testing workflow involves overcoming challenges like maintaining sample integrity, adhering to strict quality management principles, and ensuring accurate result validation. If you are striving to implement standardized procedures and rigorous quality control as outlined in the article Master Diagnostic Testing Workflow for Reliable Results you understand how critical these steps are for reliable patient outcomes.
At Centra Diagnostics in Los Angeles we specialize in advanced molecular testing toxicology blood work and COVID 19 testing. Our cutting edge technology and first in class turnaround times support your goals for precision and consistency at every stage from patient sample collection to validated result reporting. Explore how our client focused services can streamline your laboratory operations and improve healthcare decision making today.
Are you ready to enhance your diagnostic testing with trusted expertise and innovative solutions Visit Centra Diagnostics now to learn more and take advantage of our dedicated client management and comprehensive testing options. Connect with us today for reliable results that empower better healthcare decisions.
Frequently Asked Questions
What are the essential steps in the diagnostic testing workflow?
The essential steps in the diagnostic testing workflow include setting up a diagnostic testing environment, collecting and labeling patient samples, processing samples, conducting diagnostic testing, validating results, and reporting findings to healthcare providers. To ensure reliability, follow these steps systematically and implement best practices at each stage.
How can I maintain sample integrity during collection and transport?
To maintain sample integrity, follow proper identification protocols when collecting samples and use appropriate packaging during transport. Always label samples clearly with patient information, collection date, and specific test requirements before securely transporting them to the laboratory.
What quality control measures should I implement during diagnostic testing?
Implementing individualized quality control plans and regular calibration checks for diagnostic equipment helps ensure accurate testing results. Create a checklist that includes verification of reagent quality and establish performance metrics to detect any anomalies during testing.
How can I effectively validate diagnostic test results?
To validate diagnostic test results, cross-reference the findings with established control samples and baseline parameters. Perform multiple checks and statistical analysis to identify any significant deviations or errors before finalizing the results.
What should be included in a diagnostic report for healthcare providers?
A diagnostic report should include detailed test results, reference ranges, and any abnormal values, along with contextual interpretation of the findings. Make sure the report is clearly formatted and highlights any critical observations that may require prompt medical attention.
How can I ensure timely reporting of diagnostic findings?
To ensure timely reporting, develop a standardized reporting template that organizes data for quick reference, especially for urgent findings. Aim to transmit the completed report securely to healthcare providers as soon as it is validated, ideally within a few hours of completing the testing.
Recommended
- Step by Step COVID-19 Testing: Complete Guide for 2025 | Centra Diagnostics
- Diagnostics Terminology Explained: Essential Guide for 2024 | Centra Diagnostics
- 7 Top Toxicology Screening Tips for Accurate Results | Centra Diagnostics
- Guide to Ordering Blood Work: Step-by-Step for Professionals | Centra Diagnostics