Over 80 million COVID-19 tests have been performed in the United States, and every successful result depends on following each step with care. Even small mistakes in preparation or sample handling can lead to delays, inaccurate results, or unnecessary stress. Understanding each part of the testing process helps you avoid these pitfalls and ensures you get trustworthy answers when you need them most.
Table of Contents
Quick Summary
| Key Point | Explanation |
| 1. Gather all necessary materials | Collect testing swabs, containers, and identification documents to ensure smooth COVID-19 testing. |
| 2. Follow proper sample collection techniques | Ensure a high-quality sample by using sterile swabs and following instructions from healthcare professionals. |
| 3. Label specimen accurately | Clearly label with your personal information to prevent mix-ups and ensure proper processing. |
| 4. Transport sample quickly and safely | Minimize time and handle samples carefully to maintain integrity during transport to the laboratory. |
| 5. Communicate with healthcare providers | Review test results thoroughly and seek guidance from your healthcare provider for further steps based on results. |
Step 1: Prepare Necessary Materials and Documentation
Getting ready for your COVID-19 test means gathering the right supplies and paperwork to ensure a smooth testing experience. According to CDC, having the proper materials is critical for accurate specimen collection and processing.
You will need to collect several key items before your test. These typically include an approved COVID-19 testing swab, appropriate transport media, a clean collection container, and your personal identification documents. The FDA recommends using specific types of sterile swabs designed for viral testing, such as synthetic fiber swabs with thin plastic or wire shafts. Avoid cotton swabs or those with wooden handles, as these can interfere with test accuracy.
One helpful tip: Double-check all your documentation before leaving home. Ensure you have a valid photo ID, insurance card if applicable, and any pre-registration paperwork from your testing facility. This preparation will help streamline your testing process and prevent unnecessary delays or complications. Once you have everything assembled, you are ready to move forward to the actual specimen collection stage.
Step 2: Collect a High-Quality Sample Safely
Collecting a COVID-19 sample requires precision and care to ensure accurate test results. According to CDC, proper specimen collection is crucial for obtaining reliable diagnostic information and protecting both patients and healthcare providers.
The collection process depends on the specific type of test. For nasopharyngeal swab testing, you will insert a sterile swab deep into the nasal cavity, carefully rotating it to collect adequate cellular material. Research from PMC emphasizes the importance of maintaining strict biosafety protocols during this procedure. This means wearing appropriate personal protective equipment, using a gentle but firm technique, and avoiding contamination of the sample.
One critical tip: Stay completely still during the swabbing process and follow the healthcare professional’s instructions precisely. A sudden movement can compromise sample quality or cause discomfort. After collection, the sample will be carefully sealed and transported to a laboratory for processing. Your cooperation and calm demeanor will help ensure the most accurate testing experience possible.
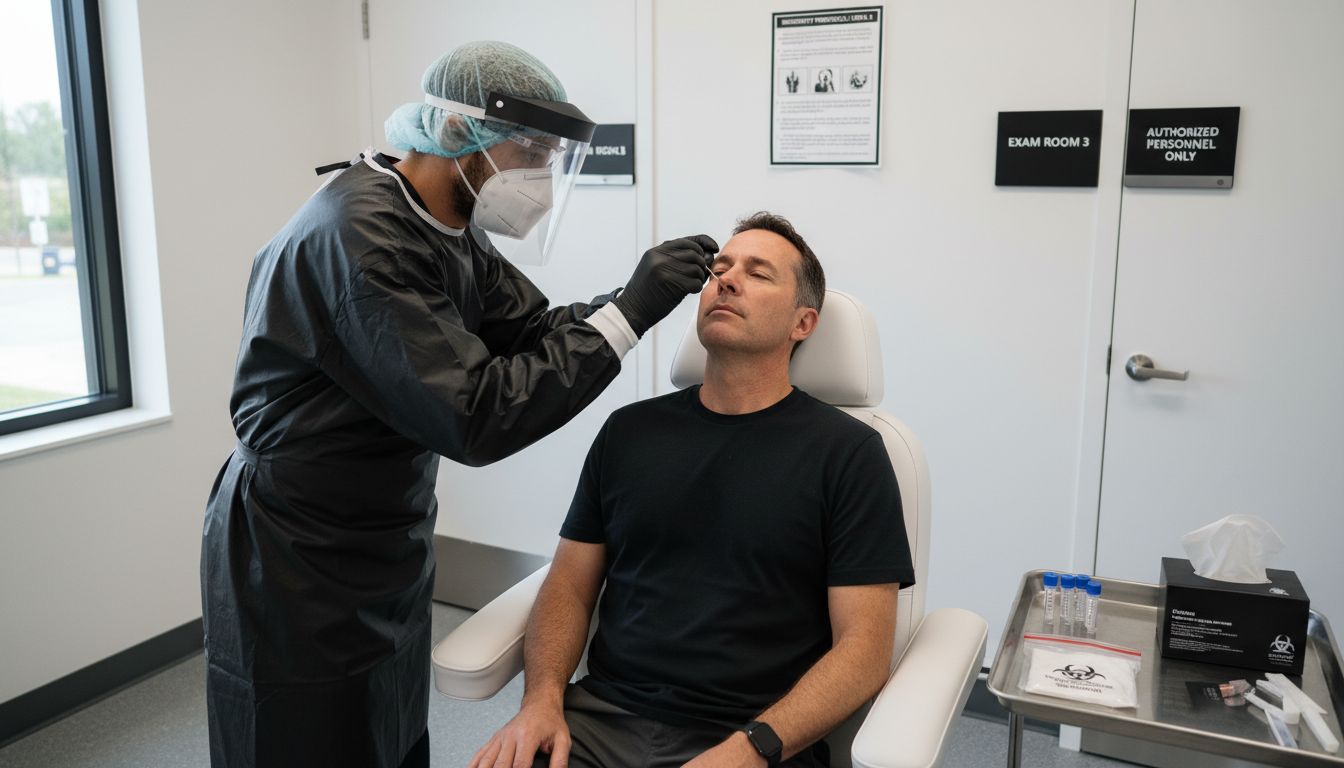
Step 3: Label and Secure the Test Specimen
After collecting your COVID-19 test sample, proper labeling and securing become critical to maintaining its integrity and ensuring accurate processing. CDC emphasizes the necessity of meticulous specimen identification and protection during transport.
The labeling process requires careful attention to detail. You will need to attach a label with your full name, date of birth, collection date, and any unique patient identifier provided by the testing facility. Research from PMC recommends using waterproof labels and placing them directly on the specimen container to prevent any potential mix-ups or information loss. Make sure the label is securely attached and the writing is clear and legible.
One crucial tip: Double-check all information before sealing the specimen container. Verify that the label matches your identification documents and that the container is completely sealed to prevent leakage. Your careful handling ensures the sample remains uncontaminated and traceable through the entire testing process. Once labeled and secured, your specimen is ready for transportation to the laboratory for analysis.
Step 4: Transport the Sample to the Laboratory
Transporting your COVID-19 test sample requires careful attention to ensure its integrity and prevent potential contamination. According to PHMSA, there are specific regulations governing the safe transportation of medical specimens.
The transportation process involves maintaining proper temperature and protection for your sample. You will need to place the sealed specimen container in a specialized biohazard transport bag or container that prevents leakage and maintains the sample’s stability. CDC recommends using insulated containers with temperature control mechanisms to preserve the sample’s diagnostic potential during transit. If you are transporting the sample yourself, keep it upright and avoid extreme temperature fluctuations.
One critical tip: Minimize the time between sample collection and laboratory delivery. The shorter the transportation time, the more accurate your test results are likely to be. If you cannot immediately transport the sample, follow the specific storage instructions provided by your healthcare professional. Some specimens may require refrigeration or specific handling to maintain their testing quality. Once you arrive at the laboratory, hand the sample directly to a laboratory technician who can immediately process and analyze your specimen.
Step 5: Verify Results and Communicate Findings
Navigating your COVID-19 test results requires careful understanding and proactive communication. FDA guidelines emphasize the critical importance of accurate result interpretation and comprehensive reporting.
When you receive your test results, carefully review all documentation provided by the testing facility. Positive results will typically include specific instructions for isolation and potential follow-up testing. Negative results might require additional context based on your symptoms and exposure history. According to FDA policies, understanding the nuanced meaning of your test result is crucial for making informed health decisions.
One critical tip: Contact your healthcare provider immediately if you have questions about your results or need guidance on next steps. They can help you interpret the test findings in the context of your personal health history and current symptoms. Remember that a single test result is just one piece of information your healthcare team will use to assess your overall health and potential COVID-19 exposure.
Ensure Accurate COVID-19 Testing with Expert Support from Centra Diagnostics
Navigating through each step of COVID-19 testing can feel overwhelming, especially when precision and safety are critical. From preparing the right materials and collecting a high-quality sample to securely transporting it and understanding your test results, every detail impacts your health decisions. At Centra Diagnostics, we understand these challenges and offer advanced COVID-19 testing solutions with first-in-class turnaround times and expert guidance to alleviate your concerns.
Experience reliable and client-focused service designed to protect your health and provide clarity when you need it most. Visit Centra Diagnostics to access comprehensive COVID-19 testing backed by state-of-the-art technology and dedicated support. Take control of your testing process today and get results you can trust with our COVID-19 testing services. Don’t wait for uncertainty to hold you back. Contact us now and move forward with confidence.

Frequently Asked Questions
What materials do I need to prepare before my COVID-19 test?
Gather essential items such as an approved COVID-19 testing swab, transport media, a clean collection container, and your personal identification documents. Confirm you have these materials ready to ensure a seamless testing process.
How do I safely collect a high-quality COVID-19 sample?
To collect a sample accurately, follow the healthcare professional’s instructions closely. Remain still and insert a sterile swab deep into the nasal cavity while rotating it, ensuring you gather ample cellular material for testing.
How should I label and secure my COVID-19 test specimen?
Carefully label your specimen container with your full name, date of birth, collection date, and any unique patient identifier. Ensure the label is waterproof, securely attached, and matches your identification documents before sealing the container.
What steps should I take to transport my COVID-19 sample to the laboratory?
Place your sealed specimen in a biohazard transport bag that prevents leakage and keeps the sample stable. Aim to deliver the sample to the laboratory as soon as possible to maintain its integrity and ensure timely testing results.
How do I verify and interpret my COVID-19 test results?
Review the documentation provided by the testing facility and understand the implications of your results. Contact your healthcare provider immediately to clarify any questions regarding your test findings and to discuss subsequent steps you may need to take.